Welcome to your Random chemistry FINAL
141. cis-trans isomerism is
125. The general formula for an ether is
167. During an exothermic reaction the compounds with less energy are:
236. If chnges in the nucleus of a chemical element occurs, the observed phenomena are:
220. Of the two pairs of acids, which one is stronger in each pair?
171. the equilibrium constant of endothermic reaction depends on:
150. The heat of formation of pure carbon in diffwrent allotropic forms:
182. What is the hybridization of each C atom in \[C_2H_4?\]
147. The rate of a chemical reaction decreases if:
153. Which type of organic compound does not contain a carbonyl group?
139. The general formula fo an ether is
207. Which of the following best describes a solution of acid?
Litmus Colour Reaction with Zn
122. Which type of organic compound does not contain a carbonyl group?
218. Which of the following is the product of the reaction of butanal with H2/Pt:
188. What are the possible oxidation states of sulphur:
157. Which one of the following bases is strong: Ca(OH)2, Al(OH)3, NH4OH, C5H5N
238. How many single electrons have a phosphorus atom in not exited state:
230. What is the next step in balancing the half reaction:
\[Cr_2O_7{^{2-}} → 2Cr^{3+}\] ?
247. Which of the statments are true?
Methane, Ethane, Hexane are:
I. homologies
II. are alcanes
III. are isomers
IV. are alkenes
154. Select the monosaccharide (s).
129. During an endothermic reaction the compounds with less energy are:
115. d-elements in the periodic table are:
174. The secondary structure of a protein is the result of _______ bonding.
146. At equilibrium, concentration is of reactants:
128. \[CH_3-CH_2-C(=O)NH_2\] is called a(n)___________
191. The chemical reaction rate is fastest when occurs between:
164. In which compound there is ionic chemical bond:
246. __________________is a monosaccharide
179. Which structure below represents a ketone?
143. Which of the properties of simple aromatic coumpound is true?
183. For which of the compounds below are cis-trans isomers possible:
204. What is the IUPAC name of the compound shown:
149. During an endothermic reaction the compounds with less energy are:
203. What is the name of the compound below:
215. Which of the following compound is NOT a monosaccharide?
160. Which of the statements are true?
I Metals have high melting and boiling points
II Metals have low boiling point
III Metals are good conductors of heat and electricity
IV Metal do not react with oxygen
186. The total sum of protons and neutrons in the nucleus of a chemical element determines:
240.The chemical reaction rate is fastest when occurs between:
199. What is the symbol for an ion which has 8 protons and 10 electrons?
239. Electron configuration of phosphorus is:
169. The rate of a first order reaction is:
118. The two molecules represented below are:
245. Which of the following compounds does not contain a - C=O bond?
194. The equilibrium constant:
166. The thermal effect of chemical reaction represents:
231. What is the next step in balancing the half reaction:
\[SO_3^{{2-}}+H_2O→SO_4^{{2-}}+2H^+\] ?
219. Soaps are:
I. lond-chain esters
II. Long-chain fatty acids
III. Sodium salts of long-chain carboxylic acids
IV. Both polar and nonpolar molecules
241. If the temperature of a chemical reaction is increased, then the equlibrium constant:
216. Consider the following statement concerning the effect of the methyl group, - CH3, on an electrophil aromatic substitution:
I. The methyl group will activate the aromatic ring.
II. The methyl group will deactivate the aromatic ring.
III The methyl group will be meta-director.
IV. The methyl group will be an ortho- and a para-director.
209. Which compound undergoes the substitution reaction with NaCN?
159. In the reaction below, what atoms or ions are being oxidized and reduced, respectively.
\[HNO_2 +\,→\,NO + I3 -\]
234. \[Ag^+_{(ag)}+I^-_{(ag)} → AgI_{(s)}\] is example of:
114. The number of atomic orbitals in the valence shell Ca atom are:
192. When temperature is decreased the rate of a chemical reaction:
225. The name for the compound with the formula
\[CH_3CH_2CH_2CH_2OH\] is
177. Proteins are biopolymers formed via multiple condensation coupling of which two functional groups?
222. What is the major functional group in the following compound?
229. What is the IUPAC name of
\[CH_3CH_2CH_2CH(CH_3)CH_3\]?
208. Which of the following is the product of the reaction of 1-hexyne with 1 mol of
211. The correct IUPAC name for the following compound is:
168. If the activation energy is decreased, the reaction rate:
175. The oxidation of ethanol produces _______.
243. The principle difference between fructose and glucose is that_______.
173. The hybridization of the central carbon atom in an aldehyde is _________.
140. _________________ is a disaccharide
232. The oxidation number of C in
170. Equilibrium can be established during:
116. How many are the acceptor atoms involved in formation of hydrogen bonds between guanine and cytosine:
217. Which statement about the carbonyl group os alderhydes is true?
I. The carbon atom in the carbonyl group can attract nucleophiles.
II. The carbon atom in the carbonyl group can attract electrophiles.
III. The carbonyl group undergoes addition reaction.
IV. The carbon atom in the carbonyl group can attract Lewis acids
144. The name of the compound
\[CH_3CH_2C(CH)_2CH_2CH_3\] is:
205. Which one of the following ions has the electron configuration
214. Which one correctly depicts a quaternary ammonium ion?
227. How many isomers are possible for dibromobenzene:
213. Peptide bonds can be classified as:
221. Sugars are examples of what type of molecule?
195. The electrons in the first electron shell have:
187. Where you can find double bond in the following six compounds:
1. NH3Cl; (2) HMnO4; (3) C2H2; (4) HClO4; (5) K2CrO4; (6) HClO;
134. The principal difference between fructose ans glucose is that__________
184. What of the non-metals is usually used to kill microorganisms in water?
176. The following reaction would produce a(n) _______.
202. In a redox reaction, there must be:
212. Which reactionis NOT possible with acetic acid?
161. Which of the statements are true?
I. oxides of metals react with water to form hydroxides
II. oxides of metals react with water to form acids
III. oxides of non-metals react with water to form acids
IV. oxides of non-metals do not react with water
- II and IV
- I and III
- I and IV
- II and IV
242. \[CH_3-CH_2-C(=O)NH_2\] is called a(n)_______________________
142. Which of the statements are true?
Methane, ethane,hexane are?
I. homologies
II. are alkanes
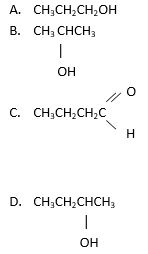
III. are isomers
IV. are alkenes
136. I, 2-dichlorethene has
156. Which of the compound reacts most readily with aqueous ammonia?
244. 1, 2-dichlorethene has
135. Which one of the following statement is true?
152. Which choice gives the structures of the reaction products when the ester below is hydrolyzed in acidic solution?
119. Which of these statements describes a condensation reaction?
165. The shared electron pair in a covalent non-polar bound spatial is localized:
145. If the concentration of a reactant is increase at constant temperature, then the equilibrium constant:
180. Which one of the following is alkyne?
196. The maximal number of electrons in the second electron shell is:
130. The heat of formation of pure carbon in different allotronic forms:
210. Which of the following alcohols gives the best yield of diakyl either by heating with a trace of sulfuric acid?
155. What is the IUPAC name of the compound shown?
127. What is the correct classification for this alcohol?
126. Which of the following is classified as an aldehyde?
162. The compounds CH3COCH2CH2CH3 and CH3CH2CH2CH2CH2OH are:
206. The net ionic equation for the hydrolysis of the salt
181. Which of the following is alkane:
148. When temperature is increased the rate increases:
131. Which compound can form hydrogen chemical bonds:
193. The equilibrium constant:
123. Select the monosaccharide (s).
190. In which row the heat of formation of the gases constantly increases:
189. During an endothermic reaction:
228. The IUPAC name of para-dibromobenzene is:
226. Markovnikov's rule is a reaction of
138. Which of the following is classified as an aldehyde?
121. The alcohol that contains two alkyl groups attached to the carbon bonded to the –OH group is a
178. Which one of the following compounds is an isomer of :
\[CH_3CH_2CH_2CH_2OH\]
235. The isotopes of a chemical element have:
172. The compound below is a(n) __________.
200. Select the formula of potassium dichromate:
201. What are the products of reaction Zn + CuSO4→ :
198. which bands are formed by a carbon atom with sp2 hybridization?
117. Is it possible to find the enthalpy of CO(g) from the reaction, if we know:
\[CO_2(g)+H_2(g) -> CO(g)+H_2O(g)+ΔH_f\]
158. Which salt when dissolved in water would form a basic solution: KClO4, KCl, CH3COOK, KNO3
185. The number of protons in the nucleus of a chemical element determines:
151. Which one correctly depicts a primary amine?
224. The reaction of an alcohol and a carboxylic acid yields
124. In the IUPAC nomenclature system, the name of which of the following would end in -al?
197. The nucleus of an atom: